Ammonium Phosphate + Sodium Sulfate
 | |
 | |
| Names | |
|---|---|
| IUPAC name ammonium phosphate | |
| Other names triammonium phosphate | |
| Identifiers | |
| CAS Number |
|
| 3D model (JSmol) |
|
| ChemSpider |
|
| ECHA InfoCard | 100.030.709 |
| EC Number |
|
| PubChem CID |
|
| UNII |
|
| CompTox Dashboard (EPA) |
|
| InChI
| |
| SMILES
| |
| Backdrop | |
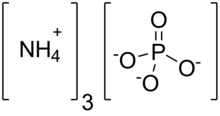
| Chemical formula | (NH4)3PO4 |
| Molar mass | 149.09 yard/mol |
| Appearance | White, tetrahedral crystals |
| Solubility in water | 58.0 thousand/100 mL (25 °C) |
| Solubility | Insoluble in acetone[2] |
| Hazards | |
| GHS labelling: | |
| Pictograms |  |
| Signal word | Warning |
| Hazard statements | H302, H319 |
| Precautionary statements | P264, P270, P280, P301+P312, P305+P351+P338, P330, P337+P313, P501 |
| NFPA 704 (burn diamond) | 2 0 0 |
| Thermochemistry | |
| Std enthalpy of | −1671.9 kJ/mol |
| Related compounds | |
| Other cations | Trisodium phosphate Tripotassium phosphate |
| Related compounds | Diammonium phosphate Monoammonium phosphate |
| Except where otherwise noted, data are given for materials in their standard country (at 25 °C [77 °F], 100 kPa). Infobox references | |
Ammonium phosphate is the inorganic compound with the formula (NH4)threePO4. Information technology is the ammonium salt of orthophosphoric acid. A related "double salt", (NH4)3PO4 .(NHiv)twoHPOiv is likewise recognized but is impractical use. Both triammonium salts evolve ammonia. In contrast to the unstable nature of the triammonium salts, the diammonium phosphate (NHiv)twoHPO4 and monoammonium table salt (NH4)HiiPOfour are stable materials that are usually used as fertilizers to provide plants with fixed nitrogen and phosphorus.[3]
Preparation of triammonium phosphate [edit]
Triammonium phosphate can exist prepared in the laboratory past treating 85% phosphoric acid with xxx% ammonia solution:[ commendation needed ]
- HthreePO4 + 3 NH3 → (NH4)3POiv
(NHiv)3POfour is a colorless, crystalline solid. The solid, which has the odor of ammonia, is readily soluble in water. The common salt converts to diammonium hydrogen phosphate (NHfour)2HPOfour.
Meet as well [edit]
- Ammonium polyphosphate
- Monoammonium phosphate
- Diammonium phosphate
References [edit]
- ^ Lide, David R. (1998). Handbook of Chemical science and Physics (87 ed.). Boca Raton, Florida: CRC Press. pp. iv–42, 5–19. ISBN978-0-8493-0594-8.
- ^ John Rumble (June 18, 2018). CRC Handbook of Chemistry and Physics (99th ed.). CRC Press. pp. 4–41. ISBN978-1138561632.
- ^ Schrödter, Klaus; Bettermann, Gerhard; Staffel, Thomas; Wahl, Friedrich; Klein, Thomas; Hofmann, Thomas (2008). "Phosphoric Acid and Phosphates". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:x.1002/14356007.a19_465.pub3.
Ammonium Phosphate + Sodium Sulfate,
Source: https://en.wikipedia.org/wiki/Ammonium_phosphate
Posted by: lewisvengland.blogspot.com



0 Response to "Ammonium Phosphate + Sodium Sulfate"
Post a Comment